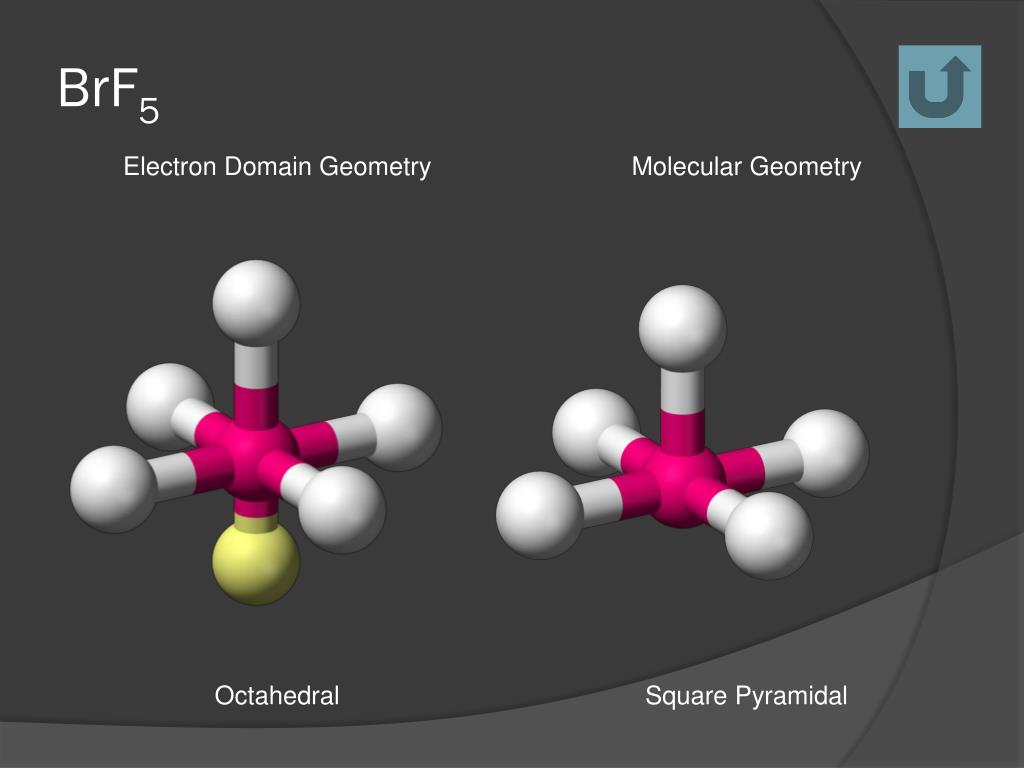
It also helps us identify the geometrical shape and the nature of the compound. The bond angle for any compound can be defined as the angle between the orbitals of the bonding pair of electrons with one central atom in a specific compound, molecule, or complex. The square pyramidal molecular geometry is a common shape for certain compounds of the main groups, which consist of active lone pairs. When we observe the shape, it describes the shapes of the compounds with the ML5 formula where L is a ligand, i.e., an ion or atom which binds the other atoms of the compound to the central atom. 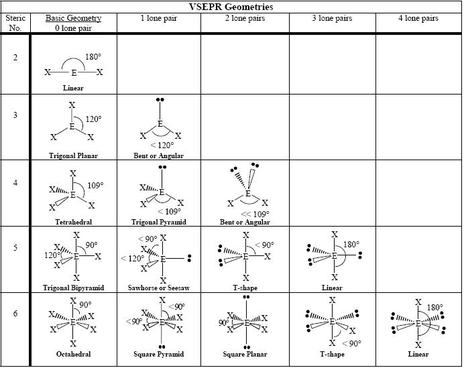
The square pyramidal shape can be defined as a molecular shape that forms when there are five bonds and one lone pair in the molecule. What is Square Pyramidal Molecular Geometry?Īs we have already discussed, the molecular geometry of BrF5 is square pyramidal. As for the molecular geometry of BrF5, it is of square pyramidal shape. It can influence some of the properties of the compound like colour, magnetism, the biological activity of the compound, reactivity, phase of matter and polarity. Here, we will focus on the molecular geometry of one specific compound, Bromine Pentafluoride. Different chemical compounds have their molecular geometry. The structure of atoms in the three-dimensional arrangement that constitute the molecules is defined as molecular geometry. Besides rocket propellants and uranium processors, it is also used for various analytical and laboratory purposes. The valence electrons present in this compound are a total of 42 in number. As for the uses of this toxic and corrosive compound, it is mostly used in rocket propellants and also for the processing of uranium. The electronic configuration for this compound can be presented as sp³d². BrF5 or Bromine PentafluorideīrF5 is an interhalogen compound, meaning that the molecules of this compound contain two or more different halogen atoms but no atoms of the elements belonging to any other group. 
The BrF5 molecular geometry is the square pyramidal shape, and the bond angles are 90° each. There is another set of properties as well, including usage, electronic configuration of the compound, molecular geometry, and bond angles. Its method of preparation includes treating bromine with fluorine in a large amount at a temperature of more than 150☌. The other properties of this compound include strong odour and colourlessness. The BrF5 or the Bromine Pentafluoride is a compound well-known for its high toxicity and corrosive nature.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
